Cell Line Development (CLD) is a crucial first step in the process of producing your recombinant proteins, biologics, or monoclonal antibodies (mAbs). It is therefore essential to choose a partner with extensive cell culture expertise and powerful gene editing technologies. Today, the cell line development field faces challenging directives, which include production of complex biologics such as engineered bispecific antibodies (BsAbs) and other difficult to express proteins.
At Hera, we focus on engineering mammalian cells–like CHO (Chinese hamster ovary) cells and HEK-293 cells–to generate high-expressing and high-titer clones that are optimized for research-grade or GMP bioproduction scale-up.
Our gene editing platform is ideal for producing your protein: whether it is for human therapeutics, animal health, diagnostic, or research applications.
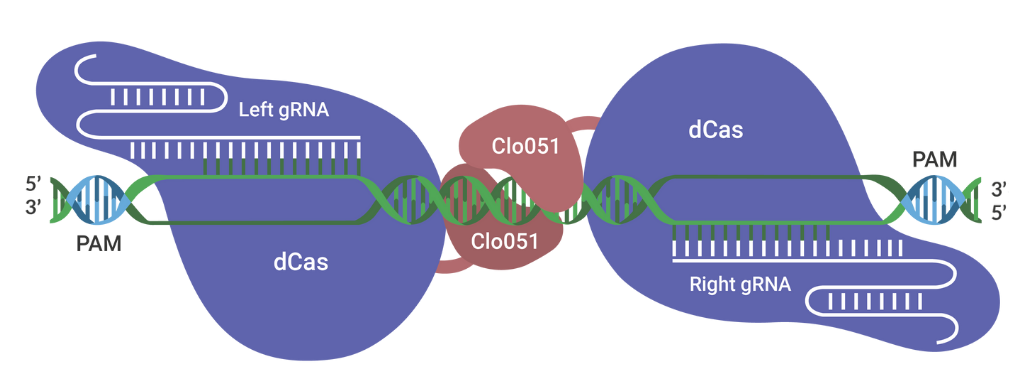
The Cas-CLOVER Site Specific Nuclease Technology is our highly specific dimeric nuclease technology for targeted gene modifications. With this technology, we engineered our GS (glutamine synthase) knockout suspension CHO cell line.
Cas-CLOVER has been optimized for high efficiency on-target nuclease activity with low to zero off target mutagenesis. The increased specificity is the result of using a pair of gRNAs and the obligate dimer Clo051 for the enzymatic activity.
We have freedom to operate using our proprietary patented Cas-CLOVER technology, allowing Hera to provide custom cell line development services with flexible and clear commercial terms.
Cell Line Development Process
Our cell line development process starts with designing and building the gene editing reagents and donor plasmid containing the protein expression construct. This is followed by transfection, GS selection, and single cell cloning.
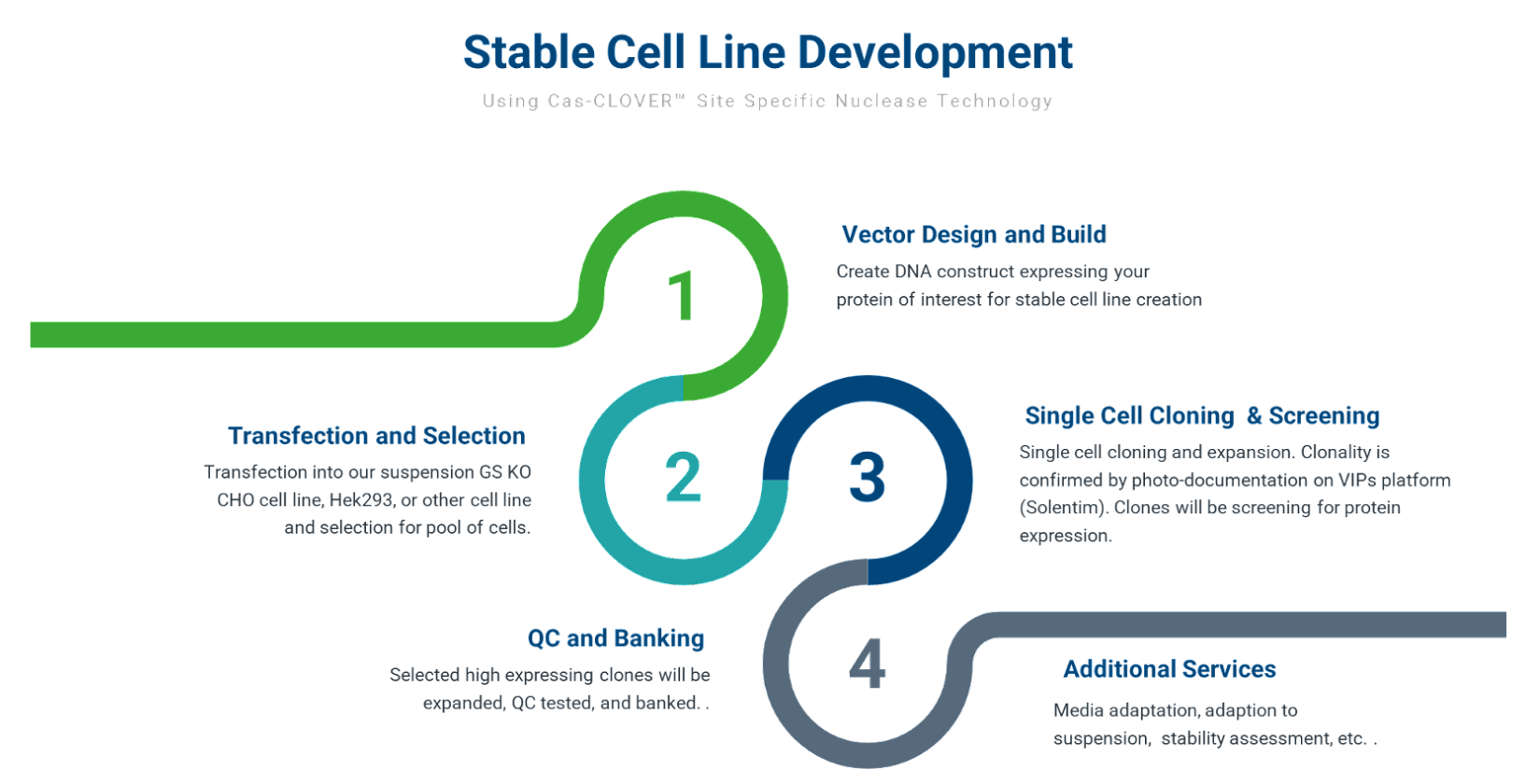
Figure 1: Overview of the Cell Line Development Process
Clones are then screened for protein expression and growth characteristics. Our process allows us to obtain accurate titer measurements and rank clone productivity. As part of our standard service offering, we provide up to 10 clones that meet client requirements. Monoclonal lines are then expanded, QC tested, and are stored as research cell banks (RCBs).
From small biotech to large pharma companies, most find the cell line development process fraught with challenges, as well as tedious and time-consuming work, which all lead to increased outsourcing at the CLD stage of protein production to specialized CROs like Hera.
Single Cell Cloning
Demonstrating that your CHO cells are monoclonal is a regulatory requirement. Hera confirms clonality with imaging/visual photo-documentation with the Solentim VIPS™ (verified in-situ plate seeding) platform designed to provide industry standard image-based evidence of clonal derivation (Figure 2). VIPS dispenses cells in nanoliter droplets into microplate wells. The cell is imaged in the bottom of the dry well as evidence of clonal seeding. Automated imaging in multiple tight z-stack layers of the whole droplet combines with intelligence-based image analysis to confirm single cell seeding. The well is then filled with growth media.
From the day of seeding and throughout the clonal growth phase, VIPS performs daily high clarity whole-well imaging, recording a timeline of well images and performing analysis to measure confluency (Figure 3). Successful clonal outgrowth can be traced back to a single cell both at the seeding event and the first whole-well image to provide dual lines of monoclonality evidence.

Figure 2: The Solentim VIPS™ Imager

Figure 3: Week 1 and 3 Clonal Outgrowth Results
Conclusion
Cell line development involves complex and dedicated processes. Outsourcing this work to well-equipped experts can vastly unburden your team.
Our Cas-CLOVER gene editing platform is more specific than CRISPR and is thus easier to use. Utilizing this technology to power our CLD services, we offer accelerated project delivery with flexible and clear commercial terms.
